Primarily the origin of electricity was based upon rotating a conductor in a magnetic field in such a way so as to cut across the lines of magnetic force.With the growing need of conservation of electricity via stored charges and to be conveniently discharged at the time of need, batteries evolved and catered to energy requirement to some extent. At that time, no one ever thought of generating electricity using any alternative technology.It was later on that eminent scientists came up with the novel idea to generate energy with help of battery via constant chargeable sources.And thus,solar cells came into existence. Though solar cells appear simple to comprehend in comparison to rotating generators, but it is actually the other way round. The principle behind electricity generation in solar cells involves light photon, ionisation and electronic battery theory.
Primarily the origin of electricity was based upon rotating a conductor in a magnetic field in such a way so as to cut across the lines of magnetic force.With the growing need of conservation of electricity via stored charges and to be conveniently discharged at the time of need, batteries evolved and catered to energy requirement to some extent. At that time, no one ever thought of generating electricity using any alternative technology.It was later on that eminent scientists came up with the novel idea to generate energy with help of battery via constant chargeable sources.And thus,solar cells came into existence. Though solar cells appear simple to comprehend in comparison to rotating generators, but it is actually the other way round. The principle behind electricity generation in solar cells involves light photon, ionisation and electronic battery theory.
To set the backdrop of the sequential explanation of our key concept, let us approach it from the basic level.
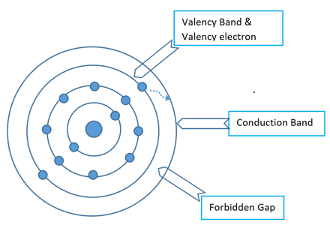
- Any atom substrate consists of nucleus and electrons moving in its orbits. In this figure,there are two electrons in the outermost orbit which are very loosely bound with nucleus.These electron scan be utilised for reactivity of that element in order make stable compounds and are known as valence electrons and the orbit is known as valency band. For conduction, electrons from outermost orbit need to jump into another orbit known as conduction band. The gap between two bands is known as forbidden gap where electrons are not allowed to stay. The size of this gap indicates the energy required for an electron to change its state from valence band to conduction band.
- The third category of elements in periodic table becomes conductive under certain suitable conditions. Silicon “Si” & Germanium “Ge” are the two main elements in this category which are utilised largely in the manufacturing of electronic components. Let us see how these elements determine its conductivity.
- To explain the conductivity scenarios across three categories of elements of periodic table, let us imagine a representation of a circular tube as a valency band and conduction band.This is just an analogy of the circular labyrinth game where balls move from one ring to another ring.
The metals have almost no band gap, that is, the valency band and conduction band overlap, so the electrons are free to jump into conduction band with slight excitement of its own energy. In semi metals some external energy is required to cross the band gap. In non-metals very high energy is required to cross the band gap, hence, this is not possible for electrons in some elements which are known as perfect insulators.
- The band structure in an atom is defined as the energy level according to the availability of electrons in that orbit. The outer most orbit of an atom contains loosely bound electrons and has the highest energy levels. The conduction band is the higher band and has no electrons, but if any kind of extra energy (light or heat) is provided to valency electrons,they get excited and jump into conduction band and become free to conduct in that material. The valency band and conduction band are separated by forbidden band and the defined energy required to jump from valency band to conduction band is known as band gap energy. This characteristic of elements differentiates metals, semi metals and non-metals.
- We all are aware that electron flow requires positive charge and negative charge, so intrinsic property of Silicon can be changed by injecting some external materials known as ‘impurities’which helps Si to become positively charged material (p-type) and negatively charged material (n-type). This is known as doping of materials. p-type semiconductor is doped with excessive positive carrier i.e. trivalent impurities are injected into intrinsic Silicon known as holes. Meanwhile,n-type semiconductor is doped with excessive negative carrier i.e. pentavalent impurities injected into intrinsic Silicon to become basic material with excessive electrons.
- In p-type semiconductors, holes are majority charge carriers while free electrons are minority charge carriers. On the other hand, in n-type semiconductors free electrons are majority charge carriers while holes are minority charge carriers. Once we join these two semiconductors, electrons start flowing in certain conditions to generate current.
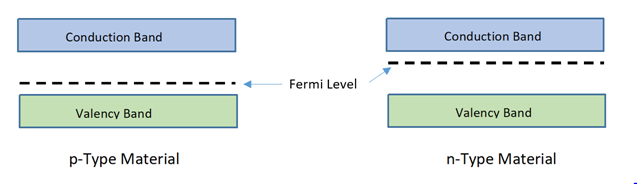
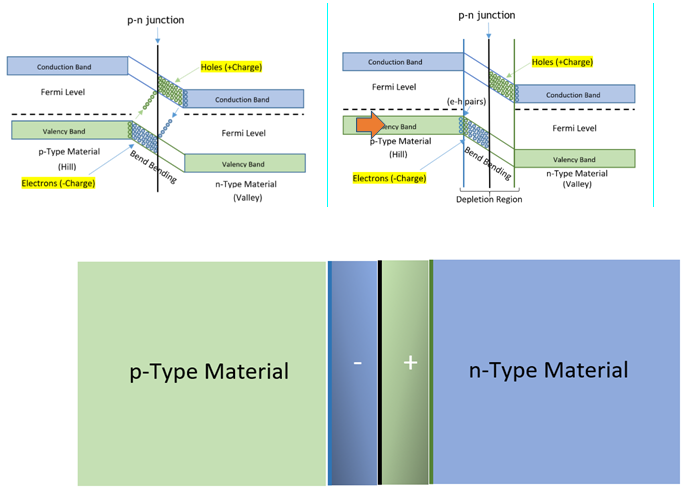
- The p-type semiconductor contains maximum positive charge in valency band and its fermi level becomes imbalanced and tends towards valency band, as greater number of holes are collected in valency band.In n-type semiconductor fermi level tends towards conduction band as high negative charge gets conducted in conduction band due to availability of high number of free electrons.
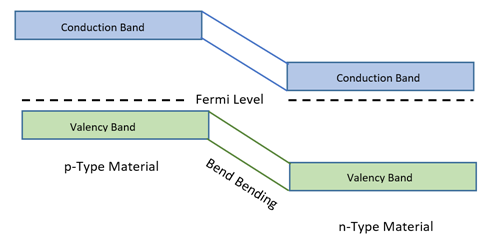
- When the combined energy levels (fermi level) of these two materials, reaches the same level at adjoining point and bends according to shift of the energy level, then the conduction band of p-type material becomes higher in level in comparison to n-type material as illustrated below. As electrons are physical particles, so considered to be the heavy particle and holes are the absence of electrons so considered to be virtual particle as light as bubble.
- Since bubbles (holes) are lighter in weight, it gets collected at the peak (valency band) of p-type material.It is difficult for these to reach thetrough of valency band of n-type material.Thus, electrons are collected at the trough of conduction band of n-type material and they cannot get to the peak of conduction band of p-type material. This situation blocks flow of electrons from one material to another material.
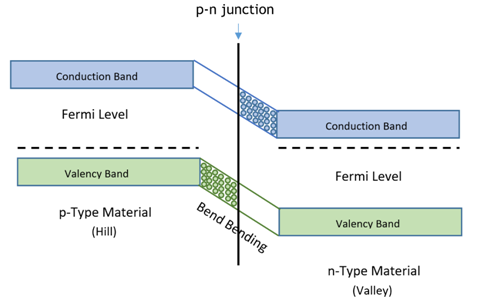
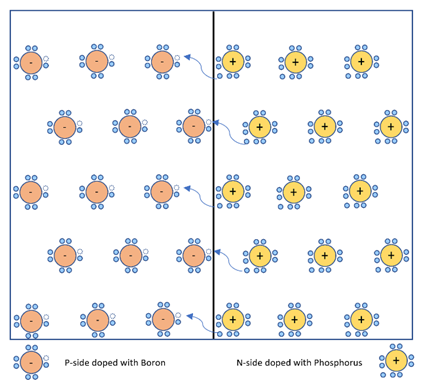
- If we join p-type semiconductor with n-type semiconductor, a p-n junction is formed. This p-n junction separates n-type semiconductor from p-type semiconductor. In n-type semiconductors, large number of free electrons are present, which being repelled from each other, try to move from a high concentration region (n-side bend bending region) to a low concentration region (p-side bend bending region). Moreover, near the junction, free electrons and holes are closer to each other. According to Coulombs law, force of attraction exists between opposite charges. Hence, free electrons from n-side gets attracted towards holes at p-side. Similarly, holes move from p-side to n-side and gets neutralised.
- To understand the function of joining of two layers, it is pertinent to know about their energy levels. Energy distribution among charged particles as described by Fermi level says, that before injecting impurities into intrinsic semiconductor, the energy level lies in between valency band and conduction band and energy in all available electrons is lower than the Fermi level. In this state the semiconductor perfectly behaves as an insulator.
- Once any extra amount of energy is provided to semiconductor, the electrons gets excited and their energy level becomes greater than Fermi level and they jump into conduction band to become free, then the semiconductor becomes conductive. To make this system, more effective, conductive, and controlled, we add some impurities into intrinsic semiconductor material, to convert them into positive and negative charge carriers as per the type of property of injected impurity material.
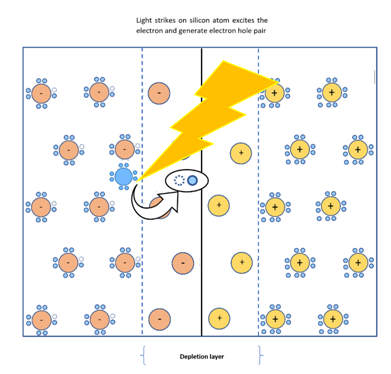
Now illustrating the complete cycle of electricity generation in solar cells sequentially with the help of a detailed diagram.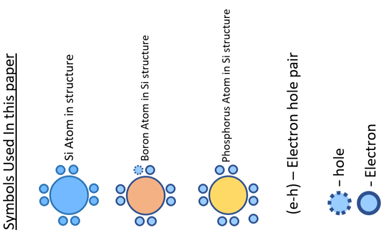
Step 1 : Intrinsic semiconductor Silicon (Si) structure
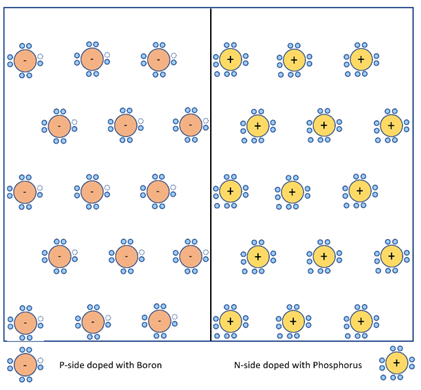
Step 2 : Intrinsic semiconductor Silicon (Si) doped with impurities, resulting in free holes added to p-side with negative nucleus, and free electrons added to n-side with positive nucleus.
Step 3 : After hiding Si atoms fromatomic structure in all these images, only impurities atoms are visible.
Step 4 : Once the p-type and n-type materials are combined, electrons of n-side are attracted by holes of p-side and cross the junction to get neutralised. This resulting in net charge, due to imbalance in number of electrons and protons present in that atom of doped material, i.e.in atomic structure of each side of the junction.
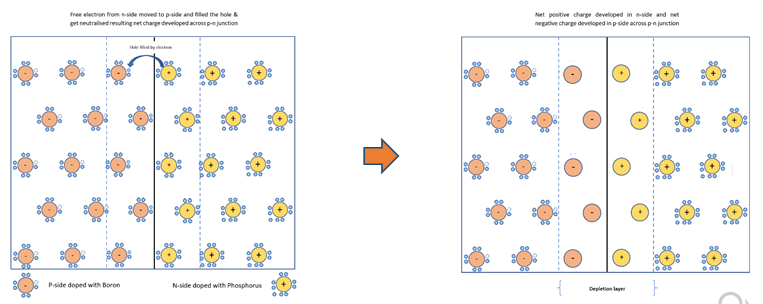
Step 5 : This process is called diffusion. The free electrons cross the junction from n-side and provide extra electrons to the atoms on p-side by filling holes in p-side atoms. The atom that gains extra electron at p-side has more number of electrons than protons. When an atom gains an extra electron from outside, it becomes a negative ion. Similarly on the other side, atom becomes positive ions. Because of ionisation net positive charge is built up towards the n-side of p-n junction and net negative charge gets built up towards p-side of the junction. This charge barrier across the p-n junction is called depletion layer and induced electric field is developed. This resembles a lightly charged battery.
Remaining free electrons and holes are prohibited to cross the junction due to charge barrier and flow within their available area.
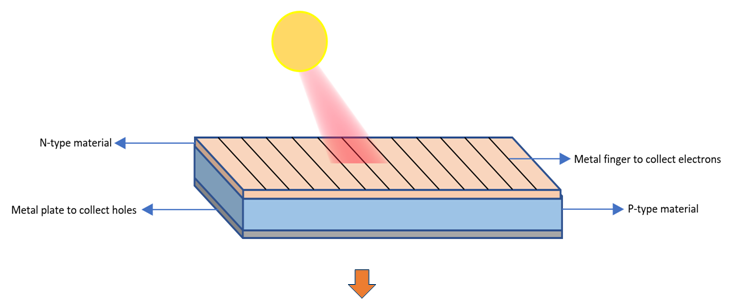
Step 6 : When this composite material is placed in sunlight and a light ray strikes at the p-n junction junction, it penetrates the layer of n-type material and transfers the photo energy to the electron of Si atom near the junction. Thus, electrons get excited and jump into conduction band.
This develops an electron-hole (e-h) pair. The separated electron from Si atom is highly energised and gets attracted by positively charged layer across the junction and moves very fast to n-type material and reaches the surface of n-type material where it is collected by metal fingers. Similarly, due to the presence of electric field, hole from same (e-h) pair moves very fast to the opposite direction of electron to meet again and gets collected by metal plate at the bottom of p-type material.
If any wire is connected across the metal finger of n-type and metal plate of p-type, current starts flowing. This phenomenon is the concept behind solar battery.
This makes solar cell act as an indefinite battery. Light incident on the solar cell surface activates electrons to flow ascurrent. Conclusively, solar cell works on three basic phenomena:
- Photon-light absorption and charge generation
- Charge separation
- Charge collection
The efficiency of any cell increases depending on the amount of charge it collects. This is how the magic of electricity generation was conceptualised without any rotation of magnetic parts.
References:
Theory of atomic structure
- Atoms are composed of nucleus which has neutrons and protons, and electrons moving in their orbits. Every atom has an equal number of protons and electrons.
- Atomic number is defined by number of protons available in the nucleus. Therefore, the periodic table is defined based upon the calculation of protons and not electrons because electrons can move out of atoms to combine with other elements to make stable compounds, but the number of protons present, are always fixed.
Periodic table is largely classified into three parts
- Metallic elements
- Non-metallic elements
- Semi metallic elements
Transition of electrons for all the three categories of elements:
From an electrical point of view, the first category of elements are conductive in nature with the range of conductivity depending upon their atomic structure e.g. gold is a better conductor than iron.
The second category of elements are non-conductive in nature i.e. they are complete insulators.
Atomic theory behind the solar cell
Once the bunch of electrons reach bend-bending region of n-type material, they are repelled by a majority of electrons present in conduction band of that material and get attracted by opposite charge holes present in bend-bending region of p-type material. As an effect of repelling and attracting forces on electrons present in bend-bending region of conduction band in n-type, they lose their energy and jump to bend-bending region of valency band in p-type, instead of climbing to bend-bending region of conduction band in p-type. Similarly, holes penetrate into bend-bending region of conduction band of n-type material. This is called the diffusion of electron holes across p-n junction. This phenomenon creates positive and negative charge across the junction due to presence of free electron and holes.
With the migration of electrons from n-side, by crossing the junction, it provides extra electrons to the atoms on p-side by neutralising holes.This creates negative ions resulting in atoms of p-type material gaining extra electrons in comparison to available protons in neutralised junction. This happens until electron gets diffused in p-type material and becomes ionised as negative and creates a negatively charged layer. Similarly, each free electron that left the atom from n-side to fill a hole in p-side atom creates a hole at n-side and neutralises electron to create a positively charged layer across the junction. This phenomenon is called diffusion and it develops the electrostatic charge across the junction.
Now when sun light strikes the junction of the cell, it excites electron of Si atom which jumps into conduction band i.e. separates from atom to conduct.In parallel a hole is also generated, and this is called (e-h) electron hole pair. This electron in conduction band of p-type material is attracted towards n-type material and because of high energy it moves with speed towards the surface of n-type of material and is collected by metal fingers. In parallel, the hole in p-side which is in pair with electron is repelled by the positive charge of n-side and moves towards the surface of p-type material and gets collected through metal plate at bottom. If there is a wire with load connected between these two,then current starts flowing.